Adoptive T-cell therapies are rapidly transforming modern medicine, offering new treatment options for cancer and other diseases. However, the manufacturing of T-cell therapies remains complex, often requiring multiple instruments, open handling steps, and manual interventions. These challenges can introduce variability, increase contamination risk, and make processes difficult to scale.
At Scinus Cell Expansion, we focus on simplifying cell therapy manufacturing. The Osilaris™ bioreactor platform enables an integrated workflow that combines T-cell selection, activation, and expansion within a single closed system, reducing manual handling while maintaining high cell quality and yield.
In this blog post, we highlight how the Osilaris platform can be used for the efficient manufacturing of therapies like CAR-T, TCR-T or Tumor Infiltrating Lymphocytes (TIL), starting directly from leukopak material.

The challenge of T-cell manufacturing
Traditional T-cell manufacturing workflows typically involve several separate process steps. Cells are often isolated, activated, and expanded using different instruments, with intermediate open event transfers between vessels.
These workflows introduce:
Process variability
Increased contamination risk
Higher Class A cleanroom footprint
Greater operational complexity
As the cell therapy field moves toward scalable manufacturing, simplifying and integrating these processes becomes increasingly important.
The Osilaris™ platform was designed to address these challenges by enabling multiple T-cell processing steps within a single closed bioreactor system.
Using the Osilaris platform, T cells can be:
Selected directly from leukopak material
Activated using TransAct™ reagents
Expanded under controlled perfusion conditions
All within a single integrated workflow.
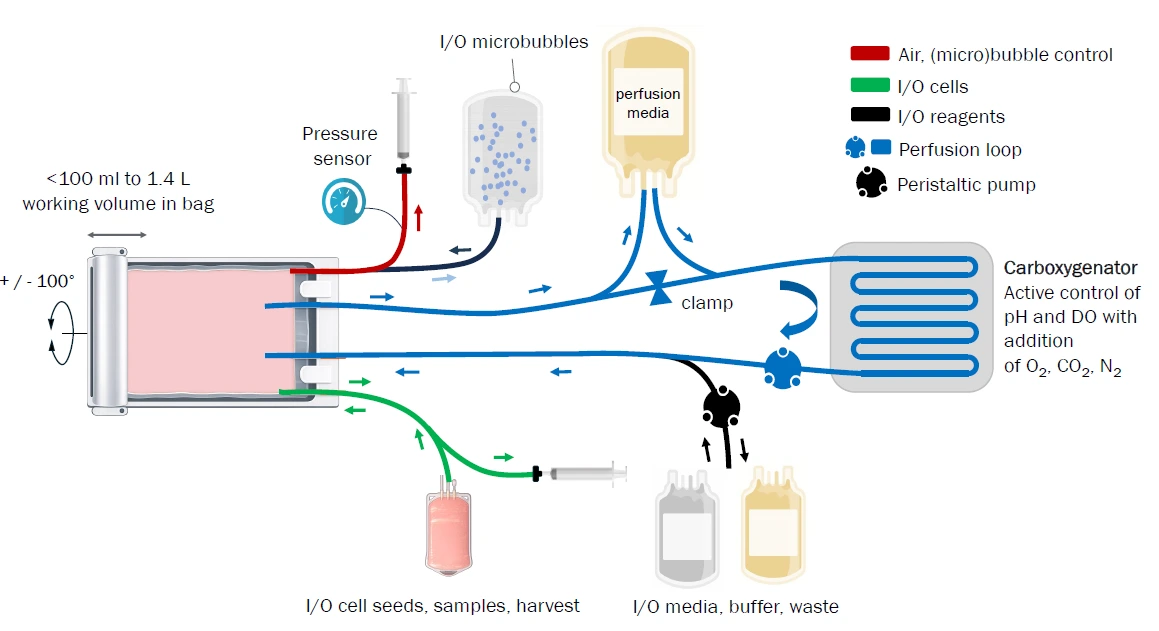
Integrated T-cell selection and expansion
The workflow begins with buoyancy-activated cell sorting (BACS™), a microbubble-based technology that enables negative selection of T cells directly within the Osilaris bioreactor.
From one-tenth of a leukopak, approximately 3–5 × 10⁸ CD3⁺ T cells can be isolated with 92–94% purity and ~98% viability.
After selection, T cells are activated and cultured directly in the Osilaris bioreactor bag, eliminating additional vessel transfers.
Key features of the system include:
1.4 L suspension culture bag
Integrated pH and dissolved oxygen sensors
Perfusion-based media exchange
Closed fluid pathway for sampling and feeding
This design allows precise control of the culture environment while maintaining a closed and GMP-compatible workflow.
Robust T-cell expansion
Activated T cells expanded for 7 days, reaching approximately 8 × 10E9 total cells by the end of culture.
This corresponds to greater than 140-fold expansion from an initial seeding of approximately 55 × 10⁶ cells.During expansion, cultures achieved cell densities exceeding 7 × 10⁶ cells/mL, demonstrating the system’s ability to support robust cell growth under perfusion conditions.
Importantly, these results were reproducible across multiple donors, highlighting the robustness of the Osilaris workflow for T-cell expansion.
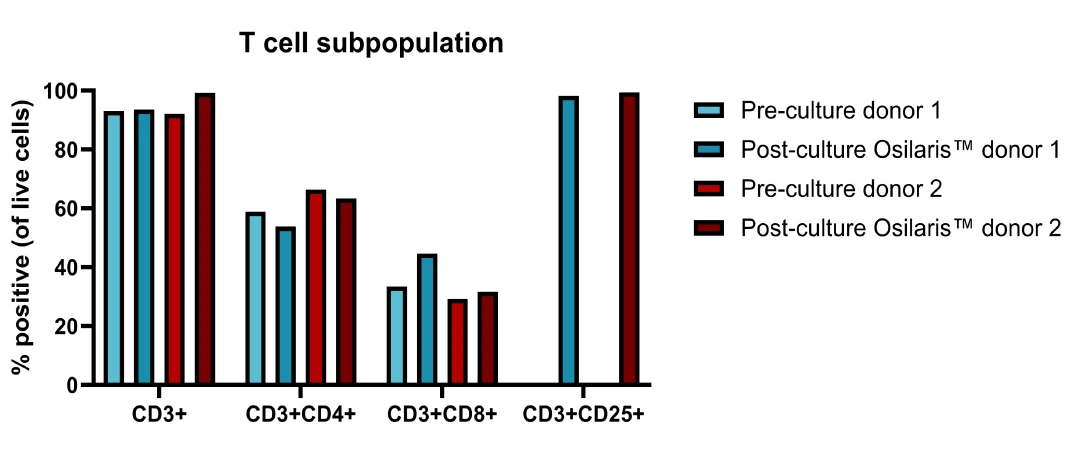
Maintaining T-cell phenotype
This indicates that the integrated workflow supports efficient expansion without skewing the T-cell population.
A streamlined solution for cell therapy manufacturing
Combining cell selection, activation, and expansion in a single platform reduces process complexity and manual handling.
Toward simpler T-cell manufacturing
As cell therapies continue to advance, manufacturing technologies must evolve to support scalable and reliable production.
The Osilaris™ bioreactor platform demonstrates how integrating T-cell selection, activation, and expansion into a single closed system can simplify workflows while maintaining strong expansion performance.
By reducing process complexity and manual handling steps, integrated bioreactor platforms may help enable more efficient and scalable T-cell manufacturing for next-generation cell therapies.